Probiotic Strain Identification: Current and Future Approaches
DOI:
https://doi.org/10.5530/ajphs.2026.16.88Keywords:
Probiotic strain identification, Strain identification approaches, ProbioticsAbstract
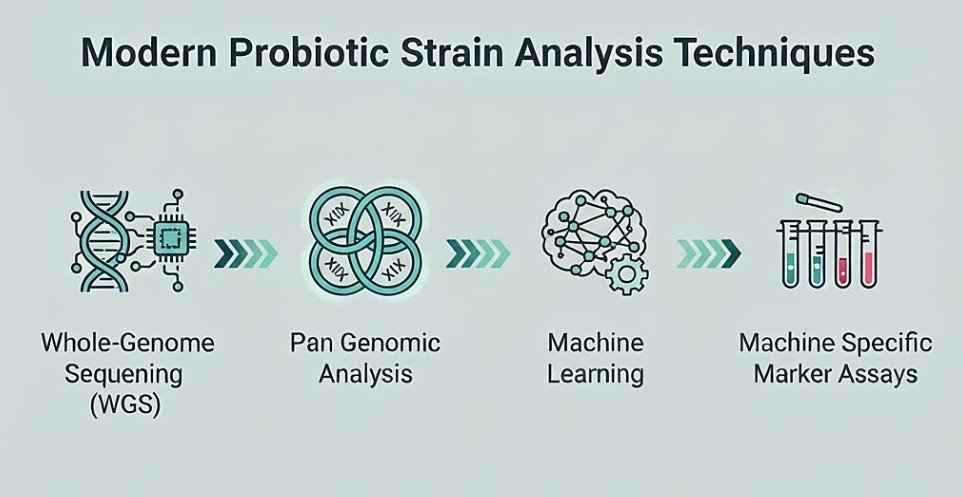
Health benefits of Probiotic are highly strain-specific, making accurate strain identification essential for safety, efficacy, correct labeling, and regulatory compliance. Over time, identification approaches have evolved from traditional culture-based and biochemical methods to advanced molecular and genomic techniques. Current methods include phenotypic and biochemical analyses, strain typing (PFGE, RAPD, MLST), mass-based fingerprinting (MALDI-TOF MS), targeted molecular assays (strain-specific PCR, qPCR, digital PCR), and high-resolution genomic tools such as 16S rRNA gene sequencing, whole-genome sequencing (WGS), and metagenomics. These techniques vary in resolution, turnaround time, cost, and suitability for clinical trials or industrial applications. Despite significant progress, challenges remain in distinguishing closely related strains, detecting low-abundance strains in complex samples, database standardization, nomenclature, and regulatory harmonization. Emerging approaches such as pangenome analysis, long-read sequencing, functional marker discovery, mobile genomic platforms, and AI-driven genomic analysis are expected to overcome current limitations and shape the future of probiotic strain identification. This review mentions the kind of extensive analysis of the present and emerging techniques of identifying the bacterial strain of probiotics, along with the technological constraints and innovative developments in the coming years in the sector.
References
Achtman, M., & Wagner, M. (2008). Microbial diversity and the genetic nature of microbial species. Nature Reviews Microbiology, 6(6), 431-440. https://doi.org/10.1038/nrmicro1872
Albanese, D., & Donati, C. (2017). Strain profiling and epidemiology of bacterial species from metagenomic sequencing. Nature Communications, 8(1), 2260. https://doi.org/10.1038/s41467-017-02209-5
Almeida, A., Mitchell, A. L., Boland, M., Forster, S. C., Gloor, G. B., Tarkowska, A., Lawley, T. D., & Finn, R. D. (2019). A new genomic blueprint of the human gut microbiota. Nature, 568(7753), 499-504. https://doi.org/10.1038/s41586-019-0965-1
Amann, R. I., Ludwig, W., & Schleifer, K. H. (1995). Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiological Reviews, 59(1), 143-169.
Binda, S., Hill, C., Johansen, E., Obis, D., Pot, B., Sanders, M. E., Tremblay, A., & Ouwehand, A. C. (2020). Criteria to Qualify Microorganisms as “Probiotic” in Foods and Dietary Supplements. Frontiers in Microbiology, Volume 11-2020. https://doi.org/10.3389/fmicb.2020.01662
Bowers, R. M., Kyrpides, N. C., Stepanauskas, R., Harmon-Smith, M., Doud, D., Reddy, T. B. K., Schulz, F., Jarett, J., Rivers, A. R., Eloe-Fadrosh, E. A., Tringe, S. G., Ivanova, N. N., Copeland, A., Clum, A., Becraft, E. D., Malmstrom, R. R., Birren, B., Podar, M., Bork, P., … The Genome Standards Consortium. (2017). Minimum information about a single amplified genome (MISAG) and a metagenome-assembled genome (MIMAG) of bacteria and archaea. Nature Biotechnology, 35(8), 725-731. https://doi.org/10.1038/nbt.3893
Boyte, M. E., Benkowski, A., Pane, M., & Shehata, H. R. (2023). Probiotic and postbiotic analytical methods: A perspective of available enumeration techniques. Frontiers in Microbiology, Volume 14-2023. https://doi.org/10.3389/fmicb.2023.1304621
Brito, P. H., Chevreux, B., Serra, C. R., Schyns, G., Henriques, A. O., & Pereira-Leal, J. B. (2018). Genetic competence drives genome diversity in Bacillus subtilis. Genome biology and evolution, 10(1), 108-124. https://doi.org/10.1093/gbe/evx270
Carr, F. J., Chill, D., & Maida, N. (2002). The Lactic Acid Bacteria: A Literature Survey. Critical Reviews in Microbiology, 28(4), 281–370. https://doi.org/10.1080/1040-840291046759
Choi, U., Park, S. H., Lee, H. B., Son, J. E., & Lee, C. R. (2023). Coordinated and Distinct Roles of Peptidoglycan Carboxypeptidases DacC and DacA in Cell Growth and Shape Maintenance under Stress Conditions. Microbiology Spectrum, 11(3), e00014-23. https://doi.org/10.1128/spectrum.00014-23
Chun, J., Oren, A., Ventosa, A., Christensen, H., Arahal, D. R., Da Costa, M. S., Rooney, A. P., Yi, H., Xu, X.-W., De Meyer, S., & Trujillo, M. E. (2018). Proposed minimal standards for the use of genome data for the taxonomy of prokaryotes. International Journal of Systematic and Evolutionary Microbiology, 68(1), 461-466. https://doi.org/10.1099/ijsem.0.002516
Dabban, I. A., Ahmad, M., Enejiyon, S. O., Hauwau, A. N., Gani, M., Oyewole, O. A., & Adetunji, C. O. (2024). Isolation techniques used for molecular characterization of beneficial microorganisms: Cultural, biochemical and molecular characterization. Hand book of Agricultural Biotechnology, 491-545.
Das, U., Mehra, R. K., Mandal, S., Ashique, S., Kumar, S., Farid, A., ... & Taghizadeh-Hesary, F. (2025). Toxicology of probiotics: Challenges and future prospects (pp. 379-428). The role of probiotics in cancer management (1st Ed).
DeMaere, M. Z., & Darling, A. E. (2019). bin3C: exploiting Hi-C sequencing data to accurately resolve metagenome-assembled genomes. Genome Biology, 20(1), 46. https://doi.org/10.1186/s13059-019-1643-1
Djukovic, A., Garzón, M. J., Canlet, C., Cabral, V., Lalaoui, R., García-Garcerá, M., Rechenberger, J., Tremblay-Franco, M., Peñaranda, I., Puchades-Carrasco, L., Pineda-Lucena, A., González-Barberá, E. M., Salavert, M., López-Hontangas, J. L., Sanz, M. Á., Sanz, J., Kuster, B., Rolain, J.-M., Debrauwer, L., … Ubeda, C. (2022). Lactobacillus supports Clostridiales to restrict gut colonization by multidrug-resistant Enterobacteriaceae. Nature Communications, 13(1), 5617. https://doi.org/10.1038/s41467-022-33313-w
Dumolin, C., Aerts, M., Verheyde, B., Schellaert, S., Vandamme, T., Van der Jeugt, F., ... & Carlier, A. (2019). Introducing SPeDE: High-throughput dereplication and accurate determination of microbial diversity from matrix-assisted laser desorption–ionization time of flight mass spectrometry data. Msystems, 4(5), 10-1128. https://doi.org/10.1128/msystems.00437-19
Felis, G. E., & Dellaglio, F. (2007). Taxonomy of Lactobacilli and Bifidobacteria. Current Issues in Intestinal Microbiology, 8(2), 44-61.
Field, D., Garrity, G., Gray, T., Morrison, N., Selengut, J., Sterk, P., Tatusova, T., Thomson, N., Allen, M. J., Angiuoli, S. V., Ashburner, M., Axelrod, N., Baldauf, S., Ballard, S., Boore, J., Cochrane, G., Cole, J., Dawyndt, P., De Vos, P., … Wipat, A. (2008). The minimum information about a genome sequence (MIGS) specification. Nature Biotechnology, 26(5), 541-547. https://doi.org/10.1038/nbt1360
Fujimoto, K., Kimura, Y., Shimohigoshi, M., Satoh, T., Sato, S., Tremmel, G., Uematsu, M., Kawaguchi, Y., Usui, Y., Nakano, Y., Hayashi, T., Kashima, K., Yuki, Y., Yamaguchi, K., Furukawa, Y., Kakuta, M., Akiyama, Y., Yamaguchi, R., Crowe, S. E., … Uematsu, S. (2020). Metagenome Data on Intestinal Phage-Bacteria Associations Aids the Development of Phage Therapy against Pathobionts. Cell Host & Microbe, 28(3), 380-389.e9. https://doi.org/10.1016/j.chom.2020.06.005
Fukao, M., Zendo, T., Inoue, T., Fuke, N., Moriuchi, T., Yamane, Y., Nakayama, J., Sonomoto, K., & Fukaya, T. (2019). Relation between cell‐bound exopolysaccharide production via plasmid‐encoded genes and rugose colony morphology in the probiotic Lactobacillus brevis KB290. Animal Science Journal, 90(12), 1575-1580. https://doi.org/10.1111/asj.13297
Gevers, D., Cohan, F. M., Lawrence, J. G., Spratt, B. G., Coenye, T., Feil, E. J., Stackebrandt, E., De Peer, Y. V., Vandamme, P., Thompson, F. L., & Swings, J. (2005). Re-evaluating prokaryotic species. Nature Reviews Microbiology, 3(9), 733-739. https://doi.org/10.1038/nrmicro1236
Gevers, D., Huys, G., & Swings, J. (2001). Applicability of rep-PCR fingerprinting for identification of Lactobacillus species. FEMS Microbiology Letters, 205(1), 31-36. https://doi.org/10.1111/j.1574-6968.2001.tb10921.x
Ghelardi, E., Mazzantini, D., Celandroni, F., Calvigioni, M., Panattoni, A., Lupetti, A., Bois De Fer, B., & Perez, M. (2023). Analysis of the microbial content of probiotic products commercialized worldwide and survivability in conditions mimicking the human gut environment. Frontiers in Microbiology, Volume 14-2023. https://doi.org/10.3389/fmicb.2023.1127321
Giraffa, G., Rossetti, L., & Neviani, E. (2000). An evaluation of chelex-based DNA purification protocols for the typing of lactic acid bacteria. Journal of Microbiological Methods, 42(2), 175-184. https://doi.org/10.1016/S0167-7012(00)00172-X
Gomaa, E. Z. (2020). Human gut microbiota/microbiome in health and diseases: A review. Antonie van Leeuwenhoek, 113(12), 2019-2040. https://doi.org/10.1007/s10482-020-01474-7
Hammes, W. P., & Vogel, R. F. (1995). The genus Lactobacillus. In B. J. B. Wood & W. H. Holzapfel (Eds.), The Genera of Lactic Acid Bacteria (pp. 19-54). Springer US. https://doi.org/10.1007/978-1-4615-5817-0_3
Han, S. S., Jeong, Y. S., & Choi, S. K. (2021). Current Scenario and Challenges in the Direct Identification of Microorganisms Using MALDI TOF MS. Microorganisms, 9(9), 1917. https://doi.org/10.3390/microorganisms9091917
Hasin, Y., Seldin, M., & Lusis, A. (2017). Multi-omics approaches to disease. Genome Biology, 18(1), 83. https://doi.org/10.1186/s13059-017-1215-1
Hill, C., Guarner, F., Reid, G., Gibson, G. R., Merenstein, D. J., Pot, B., Morelli, L., Canani, R. B., Flint, H. J., Salminen, S., Calder, P. C., & Sanders, M. E. (2014). The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nature Reviews Gastroenterology & Hepatology, 11(8), 506-514. https://doi.org/10.1038/nrgastro.2014.66
Hutkins, R. W., Krumbeck, J. A., Bindels, L. B., Cani, P. D., Fahey, G., Goh, Y. J., Hamaker, B., Martens, E. C., Mills, D. A., Rastal, R. A., Vaughan, E., & Sanders, M. E. (2016). Prebiotics: Why definitions matter. Current Opinion in Biotechnology, 37, 1-7. https://doi.org/10.1016/j.copbio.2015.09.001
Jackson, S. A., Schoeni, J. L., Vegge, C., Pane, M., Stahl, B., Bradley, M., Goldman, V. S., Burguière, P., Atwater, J. B., & Sanders, M. E. (2019). Improving End-User Trust in the Quality of Commercial Probiotic Products. Frontiers in Microbiology, 10, 739. https://doi.org/10.3389/fmicb.2019.00739
Jain, C., Rodriguez- R, L. M., Phillippy, A. M., Konstantinidis, K. T., & Aluru, S. (2018). High throughput ANI analysis of 90K prokaryotic genomes reveals clear species boundaries. Nature Communications, 9(1), 5114. https://doi.org/10.1038/s41467-018-07641-9
Janda, J. M., & Abbott, S. L. (2007). 16S rRNA Gene Sequencing for Bacterial Identification in the Diagnostic Laboratory: Pluses, Perils, and Pitfalls. Journal of Clinical Microbiology, 45(9), 2761-2764. https://doi.org/10.1128/JCM.01228-07
Jovic, D., Liang, X., Zeng, H., Lin, L., Xu, F., & Luo, Y. (2022). Single‐cell RNA sequencing technologies and applications: A brief overview. Clinical and translational medicine, 12(3), e694. https://doi.org/10.1002/ctm2.694
Kandler, O and Weiss, N (1986). Genus Lactobacillus beijerinck 1901, 212AL. In: Sneath, PHA; Mair, NS; Sharpe, ME and Holt, JG (Eds.), Bergey’s manual of systematic bacteriology. Vol. 2, Baltimore: Williams and Wilkins. PP: 1209-1234.
Lee, I. C., Tomita, S., Kleerebezem, M., & Bron, P. A. (2013). The quest for probiotic effector molecules-unraveling strain specificity at the molecular level. Pharmacological Research, 69(1), 61-74. https://doi.org/10.1016/j.phrs.2012.09.010
Libbrecht, M. W., & Noble, W. S. (2015). Machine learning applications in genetics and genomics. Nature Reviews Genetics, 16(6), 321-332. https://doi.org/10.1038/nrg3920
Liu, Y., Zheng, H., Wang, J., Xin, .J, Wu, R., Zhang, J., Zhong, Z., Liu, H., Huang, Y., Fu, H., Zhou, Z., Peng, G. (2025) Whole genome analysis and in vivo safety assessment of probiotic candidate Lactobacillus acidophilus L177. BMC Microbiol. 25(1), 398. doi: 10.1186/s12866-025-04099-w.
Liu, Y., Wang, J., Zheng, H., Xin, J., Zhong, Z., Liu, H., Fu, H., Zhou, Z., Qiu, X., Peng, G. (2024). Multi-functional properties of lactic acid bacteria strains derived from canine feces. Front Vet Sci, 11 (5), 1404580. doi: 10.3389/fvets.2024.1404580
Liu, Y., Wang, J., & Wu, C. (2022). Modulation of Gut Microbiota and Immune System by Probiotics, Pre-biotics, and Post-biotics. Frontiers in Nutrition, Volume 8-2021. https://doi.org/10.3389/fnut.2021.634897
Lugli, G. A., Mancabelli, L., Milani, C., Fontana, F., Tarracchini, C., Alessandri, G., Van Sinderen, D., Turroni, F., & Ventura, M. (2023). Comprehensive insights from composition to functional microbe-based biodiversity of the infant human gut microbiota. Npj Biofilms and Microbiomes, 9(1), 25. https://doi.org/10.1038/s41522-023-00392-6
Maiden, M. C. J., Van Rensburg, M. J. J., Bray, J. E., Earle, S. G., Ford, S. A., Jolley, K. A., & McCarthy, N. D. (2013). MLST revisited: The gene-by-gene approach to bacterial genomics. Nature Reviews Microbiology, 11(10), 728-736. https://doi.org/10.1038/nrmicro3093
Majhenič, A. Č., Lorbeg, P. M., & Treven, P. (2017). Enumeration and identification of mixed probiotic and lactic acid bacteria starter cultures. Probiotic Dairy Products, 207-251. https://doi.org/10.1002/9781119214137.ch6
Marole, T. A., Sibanda, T., & Buys, E. M. (2024). Assessing probiotic viability in mixed species yogurt using a novel propidium monoazide (PMAxx)-quantitative PCR method. Frontiers in Microbiology, Volume 15-2024. https://doi.org/10.3389/fmicb.2024.1325268
Mattarelli, P., Felis, G. E., Holzapfel, W. H., & Franz, C. M. P. (2019). International Committee on Systematics of Prokaryotes, Subcommittee on the taxonomy of Bifidobacterium, Lactobacillus and related organisms. Minutes of open and closed meetings, 19 July 2016, Dublin, Ireland. International Journal of Systematic and Evolutionary Microbiology, 69 (7), 2172-2173. https://doi.org/10.1099/ijsem.0.003351
McFarland, L. V., Evans, C. T., & Goldstein, E. J. C. (2018). Strain-Specificity and Disease-Specificity of Probiotic Efficacy: A Systematic Review and Meta-Analysis. Frontiers in Medicine, Volume 5. https://doi.org/10.3389/fmed.2018.00124
Meier-Kolthoff, J. P., Auch, A. F., Klenk, H.-P., & Göker, M. (2013). Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics, 14(1), 60. https://doi.org/10.1186/1471-2105-14-60
Morovic, W., Hibberd, A. A., Zabel, B., Barrangou, R., & Stahl, B. (2016). Genotyping by PCR and high-throughput sequencing of commercial probiotic products reveals composition biases. Frontiers in microbiology, 7, 1747. https://doi.org/10.3389/fmicb.2016.01747
Olm, M. R., Crits-Christoph, A., Bouma-Gregson, K., Firek, B. A., Morowitz, M. J., & Banfield, J. F. (2021). inStrain profiles population microdiversity from metagenomic data and sensitively detects shared microbial strains. Nature Biotechnology, 39(6), 727-736. https://doi.org/10.1038/s41587-020-00797-0
Parks, D. H., Chuvochina, M., Chaumeil, P.-A., Rinke, C., Mussig, A. J., & Hugenholtz, P. (2020). A complete domain-to-species taxonomy for Bacteria and Archaea. Nature Biotechnology, 38(9), 1079-1086. https://doi.org/10.1038/s41587-020-0501-8
Pasolli, E., De Filippis, F., Mauriello, I. E., Cumbo, F., Walsh, A. M., Leech, J., Cotter, P. D., Segata, N., & Ercolini, D. (2020). Large-scale genome-wide analysis links lactic acid bacteria from food with the gut microbiome. Nature Communications, 11(1), 2610. https://doi.org/10.1038/s41467-020-16438-8
Patro, J. N., Ramachandran, P., Barnaba, T., Mammel, M. K., Lewis, J. L., & Elkins, C. A. (2016). Culture-Independent Metagenomic Surveillance of Commercially Available Probiotics with High-Throughput Next-Generation Sequencing. mSphere, 1(2), e00057-16. https://doi.org/10.1128/mSphere.00057-16
Prosser, J. I., Bohannan, B. J. M., Curtis, T. P., Ellis, R. J., Firestone, M. K., Freckleton, R. P., … Young, J. P. W. (2007). The role of ecological theory in microbial ecology. Nature Reviews Microbiology, 5(5), 384-392. https://doi.org/10.1038/nrmicro1643
Quainoo, S., Coolen, J. P. M., Van Hijum, S. A. F. T., Huynen, M. A., Melchers, W. J. G., Van Schaik, W., & Wertheim, H. F. L. (2017). Whole-Genome Sequencing of Bacterial Pathogens: The Future of Nosocomial Outbreak Analysis. Clinical Microbiology Reviews, 30(4), 1015-1063. https://doi.org/10.1128/CMR.00016-17
Quick, J., Loman, N. J., Duraffour, S., Simpson, J. T., Severi, E., Cowley, L., Bore, J. A., Koundouno, R., Dudas, G., Mikhail, A., Ouédraogo, N., Afrough, B., Bah, A., Baum, J. H. J., Becker-Ziaja, B., Boettcher, J. P., Cabeza-Cabrerizo, M., Camino-Sánchez, Á., Carter, L. L., … Carroll, M. W. (2016). Real-time, portable genome sequencing for Ebola surveillance. Nature, 530(7589), 228-232. https://doi.org/10.1038/nature16996
Quince, C., Delmont, T. O., Raguideau, S., Alneberg, J., Darling, A. E., Collins, G., & Eren, A. M. (2017). DESMAN: A new tool for de novo extraction of strains from metagenomes. Genome Biology, 18(1), 181. https://doi.org/10.1186/s13059-017-1309-9
Richter, M., & Rosselló-Móra, R. (2009). Shifting the genomic gold standard for the prokaryotic species definition. Proceedings of the National Academy of Sciences, 106(45), 19126-19131. https://doi.org/10.1073/pnas.0906412106
Rubin, B. E., Diamond, S., Cress, B. F., Crits-Christoph, A., Lou, Y. C., Borges, A. L., Shivram, H., He, C., Xu, M., Zhou, Z., Smith, S. J., Rovinsky, R., Smock, D. C. J., Tang, K., Owens, T. K., Krishnappa, N., Sachdeva, R., Barrangou, R., Deutschbauer, A. M., … Doudna, J. A. (2022). Species- and site-specific genome editing in complex bacterial communities. Nature Microbiology, 7(1), 34-47. https://doi.org/10.1038/s41564-021-01014-7
Sabih Ur Rehman, S., Nasar, M. I., Mesquita, C. S., Al Khodor, S., Notebaart, R. A., Ott, S., ... & Alam, M. T. (2025). Integrative systems biology approaches for analyzing microbiome dysbiosis and species interactions. Briefings in Bioinformatics, 26(4), bbaf323. pphttps://doi.org/10.1093/bib/bbaf323
Salvetti, E., & O’Toole, P. W. (2017). The genomic basis of lactobacilli as health-promoting organisms. Microbiology spectrum, 5(3), 10-1128. https://doi.org/10.1128/microbiolspec.bad-0011-2016
Salvetti, E., Torriani, S., & Felis, G. E. (2012). The Genus Lactobacillus: A Taxonomic Update. Probiotics and Antimicrobial Proteins, 4(4), 217-226. https://doi.org/10.1007/s12602-012-9117-8
Sanders, M. E., Benson, A., Lebeer, S., Merenstein, D. J., & Klaenhammer, T. R. (2018). Shared mechanisms among probiotic taxa: implications for general probiotic claims. Current opinion in biotechnology, 49, 207-216. https://doi.org/10.1016/j.copbio.2017.09.007
Sarita, B., Samadhan, D., Hassan, M. Z., & Kovaleva, E. G. (2025). A comprehensive review of probiotics and human health-current prospective and applications. Frontiers in Microbiology, Volume 15-2024. https://doi.org/10.3389/fmicb.2024.1487641
Scardovi, V. (1986). Irregular non-sporulating gram positive rods. Genus Bifidobacterium Orla-Jensen 1924. Bergey's Manual of Systematic Bacterioloy, 2, 1418-1434.
Schlaberg, R., Chiu, C. Y., Miller, S., Procop, G. W., Weinstock, G., Professional Practice Committee and Committee on Laboratory Practices of the American Society for Microbiology, & Microbiology Resource Committee of the College of American Pathologists. (2017). Validation of metagenomic next-generation sequencing tests for universal pathogen detection. Archives of Pathology and Laboratory Medicine, 141(6), 776-786.
Shariat, N., & Dudley, E. G. (2014). CRISPRs: Molecular Signatures Used for Pathogen Subtyping. Applied and Environmental Microbiology, 80(2), 430-439. https://doi.org/10.1128/AEM.02790-13
Stackebrandt, E., & Goebel, B. M. (1994). Taxonomic note: a place for DNA-DNA reassociation and 16S rRNA sequence analysis in the present species definition in bacteriology. International journal of systematic and evolutionary microbiology, 44(4), 846-849. https://doi.org/10.1099/00207713-44-4-846
Sun, B. B., Chiou, J., Traylor, M., Benner, C., Hsu, Y.-H., Richardson, T. G., Surendran, P., Mahajan, A., Robins, C., Vasquez-Grinnell, S. G., Hou, L., Kvikstad, E. M., Burren, O. S., Davitte, J., Ferber, K. L., Gillies, C. E., Hedman, Å. K., Hu, S., Lin, T., … Regeneron Genetics Center. (2023). Plasma proteomic associations with genetics and health in the UK Biobank. Nature, 622(7982), 329-338. https://doi.org/10.1038/s41586-023-06592-6
Tettelin, H., & Medini, D. (Eds.). (2020). The Pangenome: Diversity, Dynamics and Evolution of Genomes. Springer International Publishing. https://doi.org/10.1007/978-3-030-38281-0
Tian, Z., Zhuang, X., Luo, M., Yin, W., & Xiong, L. (2020). The propionic acid and butyric acid in serum but not in feces are increased in patients with diarrhea-predominant irritable bowel syndrome. BMC Gastroenterology, 20(1), 73. https://doi.org/10.1186/s12876-020-01212-3
Tonkin-Hill, G., Ling, C., Chaguza, C., Salter, S. J., Hinfonthong, P., Nikolaou, E., Tate, N., Pastusiak, A., Turner, C., Chewapreecha, C., Frost, S. D. W., Corander, J., Croucher, N. J., Turner, P., & Bentley, S. D. (2022). Pneumococcal within-host diversity during colonization, transmission and treatment. Nature Microbiology, 7(11), 1791-1804. https://doi.org/10.1038/s41564-022-01238-1
Van Rossum, T., Ferretti, P., Maistrenko, O. M., & Bork, P. (2020). Diversity within species: Interpreting strains in microbiomes. Nature Reviews Microbiology, 18(9), 491-506. https://doi.org/10.1038/s41579-020-0368-1
Vandamme, P., Pot, B., Gillis, M., De Vos, P., Kersters, K., & Swings, J. (1996). Polyphasic taxonomy, a consensus approach to bacterial systematics. Microbiological Reviews, 60(2), 407-438. https://doi.org/10.1128/mr.60.2.407-438.1996
Vinderola, G., Reinheimer, J., & Salminen, S. (2019). The enumeration of probiotic issues: From unavailable standardised culture media to a recommended procedure? International Dairy Journal, 96, 58–65. https://doi.org/10.1016/j.idairyj.2019.04.010
Vos, M., & Didelot, X. (2009). A comparison of homologous recombination rates in bacteria and archaea. The ISME Journal, 3(2), 199-208. https://doi.org/10.1038/ismej.2008.93
Wayne, L. G., Brenner, D. J., Colwell, R. R., Grimont, P. A. D., Kandler, O., Krichevsky, M. I., ... & Truper, H. G. (1987). Report of the ad hoc committee on reconciliation of approaches to bacterial systematics. International journal of systematic and evolutionary microbiology, 37(4), 463-464. https://doi.org/10.1099/00207713-37-4-463
Wick, R. R., Judd, L. M., & Holt, K. E. (2023). Assembling the perfect bacterial genome using Oxford Nanopore and Illumina sequencing. PLOS Computational Biology, 19(3), e1010905. https://doi.org/10.1371/journal.pcbi.1010905
Wilkinson, M. G. (2018). Flow cytometry as a potential method of measuring bacterial viability in probiotic products: A review. Trends in Food Science & Technology, 78, 1-10. https://doi.org/10.1016/j.tifs.2018.05.006
Wilkinson, M. D., Dumontier, M., Aalbersberg, I. J., Appleton, G., Axton, M., Baak, A., ... & Mons, B. (2016). The FAIR Guiding Principles for scientific data management and stewardship. Scientific Data, 3(1), 1-9. doi:10.1038/sdata.2016.18
Woese, C. R. (1987). Bacterial evolution. Microbiological Reviews, 51(2), 221-271. https://doi.org/10.1128/mr.51.2.221-271.1987
Ye, Q., Sun, S., Deng, J., Chen, X., Zhang, J., Lin, S., Du, H., Gao, J., Zou, X., Lin, X., Cai, Y., & Lu, Z. (2023). Using 16S rDNA and metagenomic sequencing technology to analyze the fecal microbiome of children with avoidant/restrictive food intake disorder. Scientific Reports, 13(1), 20253. https://doi.org/10.1038/s41598-023-47760-y
Zawistowska-Rojek, A., Zaręba, T., & Tyski, S. (2022). Microbiological Testing of Probiotic Preparations. International Journal of Environmental Research and Public Health, 19(9), 5701. https://doi.org/10.3390/ijerph19095701
Zheng J, Wittouck S, Salvetti E, et al. (2020). A taxonomic note on the genus Lactobacillus: Description of 23 novel genera, emended description of the genus Lactobacillus Beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. International Journal of Systematic and Evolutionary Microbiology,70(4), 2782-2858. DOI: 10.1099/ijsem.0.004107