In-silico design and network pharmacological approach on schiff base derivatives with sulfonamide moieties for antimicrobial property
DOI:
https://doi.org/10.5530/ajphs.2026.16.86Keywords:
In-silico design, Sulfonamides, Schiff bases, Antimicrobial Property, Docking, Network PharmacologyAbstract
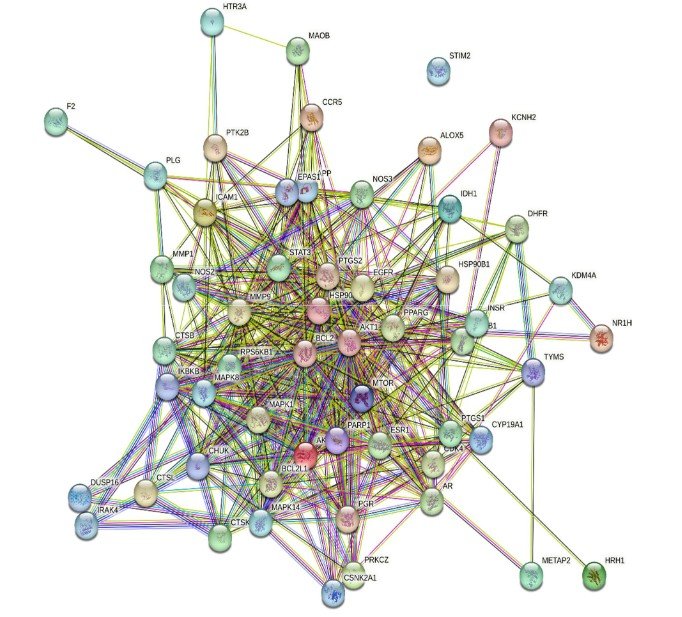
Background: Antimicrobial resistance is a global public health and economic threat. Sulfonamides are structural analogues of para-amino benzoic acid, which act as competitive inhibitors of folic acid metabolism. Although resistance has limited their use as monotherapy, sulfonamide-containing conjugates and hybrids remain valuable. Schiff bases are versatile groups with broad-spectrum biological activities, including antibacterial and antifungal properties. Objective: To design and evaluate novel Schiff base derivatives incorporating sulfonamide moieties as potential antibacterial and antifungal agents using a comprehensive in-silico drug discovery approach. Methods: A series of sulfonamide-linked Schiff bases was designed using ACD/Labs ChemSketch 12.0. All compounds were filtered according to Lipinski’s rule of five and physicochemical properties. Bioactivity scores were predicted using Molinspiration and PASS Online. ADMET profiles were assessed with admetSAR and ADMETlab2.0. Target fishing and network pharmacology analysis were performed using Swiss Target Prediction, MolsoftL.L.C, String, Gene card Venny 2.0, and Cytoscape. Molecular docking studies were carried out using Biovia Discovery Studio to evaluate binding affinity against relevant microbial targets. Results: This study has predicted the biological action of some of the proposed analogues, including the compound SB8 (4-{(Z)-[(4'-hydroxy[1,1'-biphenyl]-4-yl) methylidene] amino} benzene-1 sulfonamide), which exhibits antifungal activity and a high binding affinity for the target with a LibDock score of 93.17 and compound SB5 (4-{(E)-[(4-methyl-1H-imidazol-5-yl)methylidene]amino}benzene-1-sulfonamide) shows antibacterial activity and a binding affinity score of 71.72. Conclusion: The result showed that ligand SB5 and SB8 have high binding affinity (LibDock score: 71.72 and 71.18) to the target compared to other ligands for antibacterial activity, and ligand SB8 has high binding affinity (LibDock score: 93.17) to the target compared to other ligands for antifungal activity.
References
Al-Masoudi, W. A., Faaz, R. A., Al-Asadi, R. H., & Jabbar, H. S. (2016). Synthesis, antimicrobial activity and modelling studies of some new metal complexes of Schiff base derived from sulphonamide drug in vitro. European Journal of Chemistry, 7(1), 102-106. https://doi.org/10.5155/eurjchem.7.1.102-106.1374
Alzarea, S. I., Qasim, S., Uttra, A. M., Khan, Y. H., Aljoufi, F. A., Ahmed, S. R., ... & Malhi, T. H. (2022). Network Pharmacology and Molecular Docking Based Prediction of Mechanism of Pharmacological Attributes of Glutinol. Processes, 10(8), 1492. https://doi.org/10.3390/pr10081492
Barman, K., Das, G., G. I., Kalita, J. M., & Dasgupta, P. Design and molecular docking of sulfonamide derivatives. International Journal of Current Pharmaceutical Research, 13(4), 93-97. https://dx.doi.org/10.22159/ijcpr.2021v13i4.42752
Başaran, E., Çakmak, R., Türkmenoğlu, B., Akkoc, S., & Köprü, S. (2025). Synthesis of Sulfonamide‐Based Schiff Bases as Potent Anticancer Agents: Spectral Analyses, Biological Activity, Molecular Docking, ADME, DFT, and Pharmacophore Modelling Studies. Chemistry & Biodiversity, 22(2), e202402229. https://doi.org/10.1002/cbdv.202402229
Berredjem, M., Bouchareb, F., Djouad, S. E., Bouasla, R., Bahadi, R., Redjemia, R., ... & Dekir, A. (2023). Recent Progress in Synthesis of Sulfonamides and N‐Acylsulfonamides, Biological Applications and Their Structure‐Activity Relationship (SAR) Studies. Chemistry Select, 8(35), e202301859. https://doi.org/10.1002/slct.202301859
Bertagnolio, S., Dobreva, Z., Centner, C. M., Olaru, I. D., Donà, D., Burzo, S., …& Van Weezenbeek, K., (2024). WHO global research priorities for antimicrobial resistance in human health. The Lancet Microbe, 5(11), 100902. https://doi.org/10.1016/S2666-5247(24)00134-4
Chohan, Z. H., & Shad, H. A. (2008). Structural elucidation and biological significance of 2-hydroxy-1-naphthaldehyde derived sulfonamides and their first row d-transition metal chelates. Journal of Enzyme Inhibition and Medicinal Chemistry, 23(3), 369-379. https://doi.org/10.1080/14756360701585692
Chohan, Z. H., & Shad, H. A. (2012). Metal-based new sulfonamides: Design, synthesis, antibacterial, antifungal, and cytotoxic properties. Journal of Enzyme Inhibition and Medicinal Chemistry, 27(3), 403-412. https://doi.org/10.3109/14756366.2011.593515
Fields, U. A. P. (2012). MolSoft ICM Quarterly.
Gahimbare, L., Muvunyi, C. M., Guessennd, N. A. K., Rutanga, J. P., Gashema, P., Fuller, W., ... & Yahaya, A. A. (2024). Antimicrobial resistance in the WHO African Region: A systematic literature review 2016–2020. Antibiotics, 13(7), 659. https://doi: 10.3390/antibiotics13070659
Gao, Y. D., & Huang, J. F. (2011). An extension strategy of Discovery Studio 2.0 for non-bonded interaction energy automatic calculation at the residue level. Zoological Research, 32(3), 262-266. https://doi.org/10.3724/SP.J.1141.2011.03262
Gfeller, D., Grosdidier, A., Wirth, M., Daina, A., Michielin, O., & Zoete, V. (2014). SwissTargetPrediction: a web server for target prediction of bioactive small molecules. Nucleic Acids Research, 42(W1), W32-W38. https://doi.org/10.1093/nar/gku293
Harutyunyan, A., Gukasyan, G. T., Grigoryan, A. S., & Stepanyan, H. M. (2021). Antibacterial and mao-inhibiting properties of new styryl derivatives of pyrimidines, quinazolines and bis-quinazolines. Ajastan Kensabanakan Handes, 73(2), 26-30.
Khan, M. N., Khan, A. M., Ullah, H., Hussain, S., & Khattak, A. K. (2018). Synthesis and antibacterial activity of the sulfonamide based schiff base and its transition metal (II) complexes. Pakistan Journal of Pharmaceutical Sciences,31(1), 103-111.
Kose, L. P., Gulcin, İ., Yıldırım, A., Atmaca, U., Çelik, M., Alwasel, S. H., & Supuran, C. T. (2016). The human carbonic anhydrase isoenzymes I and II inhibitory effects of some hydroperoxides, alcohols, and acetates. Journal of Enzyme Inhibition and Medicinal Chemistry, 31(6), 1248-1253. https://doi.org/10.3109/14756366.2015.1120723
Krátký, M., Dzurková, M., Janoušek, J., Konečná, K., Trejtnar, F., Stolaříková, J., & Vinšová, J. (2017). Sulfadiazine salicylaldehyde-based Schiff bases: Synthesis, antimicrobial activity and cytotoxicity. Molecules, 22(9), 1573. https://doi.org/10.3390/molecules22091573
Lagunin, A., Stepanchikova, A., Filimonov, D., & Poroikov, V. (2000). PASS: prediction of activity spectra for biologically active substances. Bioinformatics, 16(8), 747-748. https://doi.org/10.1093/bioinformatics/16.8.747
Mondal, S., Mandal, S. M., Mondal, T. K., & Sinha, C. (2017). Spectroscopic characterization, antimicrobial activity, DFT computation and docking studies of sulfonamide Schiff bases. Journal of Molecular Structure, 1127, 557-567. http://dx.doi.org/10.1016/j.molstruc.2016.08.011
Niazi, S. K., & Mariam, Z. (2023). Computer-aided drug design and drug discovery: a prospective analysis. Pharmaceuticals, 17(1), 22. https://doi.org/10.3390/ph17010022
Otani, K. (2003). Cytochrome P450 3A4 and benzodiazepines. Seishin Shinkeigaku Zasshi, 105(5), 631-642.
Othman, R. M., Al-Masoudi, W. A., Hama, A. A., & Hussain, S. M. (2019). Antimicrobial activity and molecular modeling study of Schiff base derived from sulfamerazine. Indian Journal of Forensic Medicine & Toxicology, 13(4), 694-699.
Podos, S. D., Thanassi, J. A., & Pucci, M. J. (2012). Mechanistic assessment of DNA ligase as an antibacterial target in Staphylococcus aureus. Antimicrobial Agents and Chemotherapy, 56(8), 4095-4102. https://doi.org/10.1128/AAC.00215-12
RCSB Protein Data Bank. (n.d.). RCSB.org. https://www. rcsb. org/(visited on 09/15/2022).
Safran, M., Dalah, I., Alexander, J., Rosen, N., & Lancet, D. (2010). GeneCards Version 3:the human gene integrator. Database, 2010. https://doi.org/10.1093/database/baq020
Saito, R., Smoot, M. E., Ono, K., Ruscheinski, J., Wang, P. L., Lotia, S., ... & Ideker, T. (2012). A travel guide to Cytoscape plugins. Nature Methods, 9(11), 1069-1076. https://doi.org/10.1038/nmeth.2212
Shallangwa, G. A., Musa, H., & Ogbe, E. (2015). Design and synthesis of new series of one pot Schiff bases of 4-aminobenzenesulfon-amideas potent antibacterial and anti-fungal agents. Journal of Progressive Research in Chemistry, 1, 14-21.
Tahriri, M., Yousefi, M., Mehrani, K., Tabatabaee, M., & Ashkezari, M. D. (2017). Synthesis, characterization and antimicrobial activity of two novel sulfonamide Schiff base compounds. Pharmaceutical Chemistry Journal, 51(5), 425-428. https://doi.org/10.1007/s11094-017-1626-z
Thangavelu, P., & Thangavel, S. (2018). Design, synthesis, and docking of sulfadiazine Schiff base scaffold for their potential claim as Inha enoyl-(acyl-carrier-protein) reductase inhibitors. Asian Journal of Pharmaceutical and Clinical Research, 11(10), 233. http://dx.doi.org/10.22159/ajpcr.2018.v11i10.27179
Ul-Hassan, M., Chohan, Z. H., Scozzafava, A., & Supuran, C. T. (2004). Carbonic anhydrase inhibitors: Schiff's bases of aromatic and heterocyclic sulfonamides and their metal complexes. Journal of Enzyme Inhibition and Medicinal Chemistry, 19(3), 263-267. https://doi.org/10.1080/14756360410001689595
Xiong, G., Wu, Z., Yi, J., Fu, L., Yang, Z., Hsieh, C., ... & Cao, D. (2021). ADMETlab 2.0: an integrated online platform for accurate and comprehensive predictions of ADMET properties. Nucleic acids research, 49(W1), W5-W14. https://doi.org/10.1093/nar/gkab255
Yang, H., Lou, C., Sun, L., Li, J., Cai, Y., Wang, Z., ... & Tang, Y. (2019). ADMETSAR 2.0: web-service for prediction and optimization of chemical ADMET properties. Bioinformatics, 35(6), 1067-1069. https://doi.org/10.1093/bioinformatics/bty707
Zemede, Y. B., Nithyakalyani, D., & Kumar, S. A. (2015). Synthesis, characterization and antimicrobial properties of some transition metal complexes with NS-chelating Schiff base ligand incorporating thiophene and sulfonamide moieties. Asian Journal of Chemistry, 27(3), 941-948. http://dx.doi.org/10.14233/ajchem.2015.17627
Zerrouki, S., Bouchoucha, A., Djellouli, F., Bouzaheur, A., Si Larbi, K., & Bourouai, M. A. (2024). New Schiff base Derivatives Bearing Sulfonamide Moiety: Synthesis, In vitro Antimicrobial Activity, DFT Calculations, ADMET and Molecular Docking Study. Iranian Journal of Chemistry and Chemical Engineering, 43(1).204-224. https://doi.org/10.30492/ijcce.2023.1987960.5829